Professional Documents
Culture Documents
Ejercicio Brenda
Uploaded by
Anonymous smFxeArJL0 ratings0% found this document useful (0 votes)
3 views1 pageOriginal Title
ejercicio brenda.docx
Copyright
© © All Rights Reserved
Available Formats
DOCX, PDF, TXT or read online from Scribd
Share this document
Did you find this document useful?
Is this content inappropriate?
Report this DocumentCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
0 ratings0% found this document useful (0 votes)
3 views1 pageEjercicio Brenda
Uploaded by
Anonymous smFxeArJLCopyright:
© All Rights Reserved
Available Formats
Download as DOCX, PDF, TXT or read online from Scribd
You are on page 1of 1
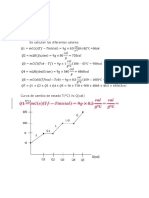
DATOS:
g NaOH =?
8 g Na pureza = 57%
8 g H2O
%R= 71%
Na + H2O → NaOH + H2
SOLUCIÓN :
8 g Na * 71g /100g = 5.68 g Na
Se balancea la reacción :
2Na + 2 H20 → 2 NaOH + H2
2 mol 2 mol 2 mol 1 mol
2 *23g 2*18 g 2*40g 2g
46 g 36 g 80 g 2g
Ahora se calcula reactivo limitante y exceso :
8 g Na * 36 gH2O/ 46 g Na = 6.26 g Na
8 g H2O * 46 gNa / 36 g H2O= 10.22 g H2O .
El H2O es el reactivo limitante y el Na es el reactivo en exceso
8 g H2O * 80 gNaOH/ 36 gH2O= 17.77 g NaOH RT
%R = RR/RT*100
Se despeja RR:
RR= %R* RT/100
RR= 71 * 17.77 NaOH/100= 12.61 g NaOH
You might also like
- Modelo de Preinforme VDocument2 pagesModelo de Preinforme VAnonymous smFxeArJLNo ratings yet
- Formato Tarea 1 Ejercicio 1Document10 pagesFormato Tarea 1 Ejercicio 1Anonymous smFxeArJLNo ratings yet
- Ejercicio1 Pretarea JuliethBarahonaDocument3 pagesEjercicio1 Pretarea JuliethBarahonaAnonymous smFxeArJLNo ratings yet
- Inform EsDocument1 pageInform EsAnonymous smFxeArJLNo ratings yet
- Tarea de MicrosDocument7 pagesTarea de MicrosHenry Alan Bolivar Toro0% (2)
- Trabajo 1Document11 pagesTrabajo 1Anonymous smFxeArJLNo ratings yet
- Anexo 4Document9 pagesAnexo 4dannyNo ratings yet
- Universidad Nacional Abierta y A DistanciaDocument5 pagesUniversidad Nacional Abierta y A DistanciaAnonymous smFxeArJLNo ratings yet
- Brenda QuimicaDocument9 pagesBrenda QuimicaAnonymous smFxeArJLNo ratings yet
- Guía de Actividades y Rúbrica de Evaluación - Paso 2 - Elaborar Estudio de Caso Bacteriemia Por SDocument7 pagesGuía de Actividades y Rúbrica de Evaluación - Paso 2 - Elaborar Estudio de Caso Bacteriemia Por SJorge Cano PeñaNo ratings yet
- Anexo 5.1-Formato Preinformes - Química OrgánicaDocument3 pagesAnexo 5.1-Formato Preinformes - Química OrgánicaDiego VacaNo ratings yet
- Anexo 5.2-Formato Informes - Química OrgánicaDocument3 pagesAnexo 5.2-Formato Informes - Química Orgánicamilena rojasNo ratings yet
- Anexo AnalisisDocument4 pagesAnexo AnalisisAnonymous smFxeArJLNo ratings yet
- Guía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Document7 pagesGuía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Anonymous smFxeArJLNo ratings yet
- Guía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Document1 pageGuía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Anonymous smFxeArJLNo ratings yet
- BrendaDocument1 pageBrendaAnonymous smFxeArJLNo ratings yet
- EnsayoDocument3 pagesEnsayoAnonymous bSdau3No ratings yet
- Trabajo 1Document10 pagesTrabajo 1Anonymous smFxeArJLNo ratings yet
- Trabajo 1Document10 pagesTrabajo 1Anonymous smFxeArJLNo ratings yet
- Guía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Document7 pagesGuía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Anonymous smFxeArJLNo ratings yet
- Guía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Document7 pagesGuía de Actividades y Rúbrica de Evaluación - Tarea 1 - Realizar Trabajo Sobre El Reconocimiento Del Curso.Anonymous smFxeArJLNo ratings yet